The $2.3 Billion Paradox: A Strategic White Paper for Executive Leadership and Venture Capitalists - I
February 12, 2026
Foreword
“The $2.3 Billion Paradox” is a three-episode strategic diagnostic for the leaders currently architecting the next century of medicine. This is not another high-level summary of R&D trends; it is a forensic audit of an industry currently strangling itself with its own systemic inefficiencies. While the “AI Revolution” promises a digital alchemy that turns data into cures, the cold, hard numbers tell a different story of skyrocketing costs and stagnant cycle times. By interpreting the data behind Eroom’s Law, this white paper strips away the “AI frenzy” to provide executive leadership and venture capitalists with a pragmatic roadmap for Engineering the Future of Biopharma R&D. We move past the “magic wand” marketing to define what it actually takes to win in a $2.3 billion reality.
Episode I:
The Reality Check — Why “Magic Wands” Fail

We’ve all heard the pitch: “AI will cut drug discovery costs in half tomorrow.” It sounds great on a slide deck, but if you look at the balance sheets of Big Pharma, the numbers tell a different story. Let’s look at the data and figure out why the “magic wand” is currently hitting a brick wall.
The Elephant in the Room: Is AI Frenzy Just Following the Herd?
Two years ago, “Generative AI” was a sci-fi buzzword. Today, it’s the air we breathe. Biopharma boards are pouring billions into AI-driven investment and baking these costs into their five-year budgets. But here is the question I’ve been asked behind closed doors:
Is this trailblazing innovation, or are we just following the herd?
To answer that, we have to look at the “Capital Inefficiency” currently strangling the industry.
The Reality Check: The “ROI Paradox”
If you assumed that more technology equals cost and time reduction, you’re in for a shock. We are currently living through “Eroom’s Law” (that’s “Moore’s Law” spelled backward)—where despite exponential tech growth, drug development is getting slower and more expensive. Take a look at the data from major consulting firms like Deloitte and research groups like Tufts Center for the Study of Drug Development (Table 1):
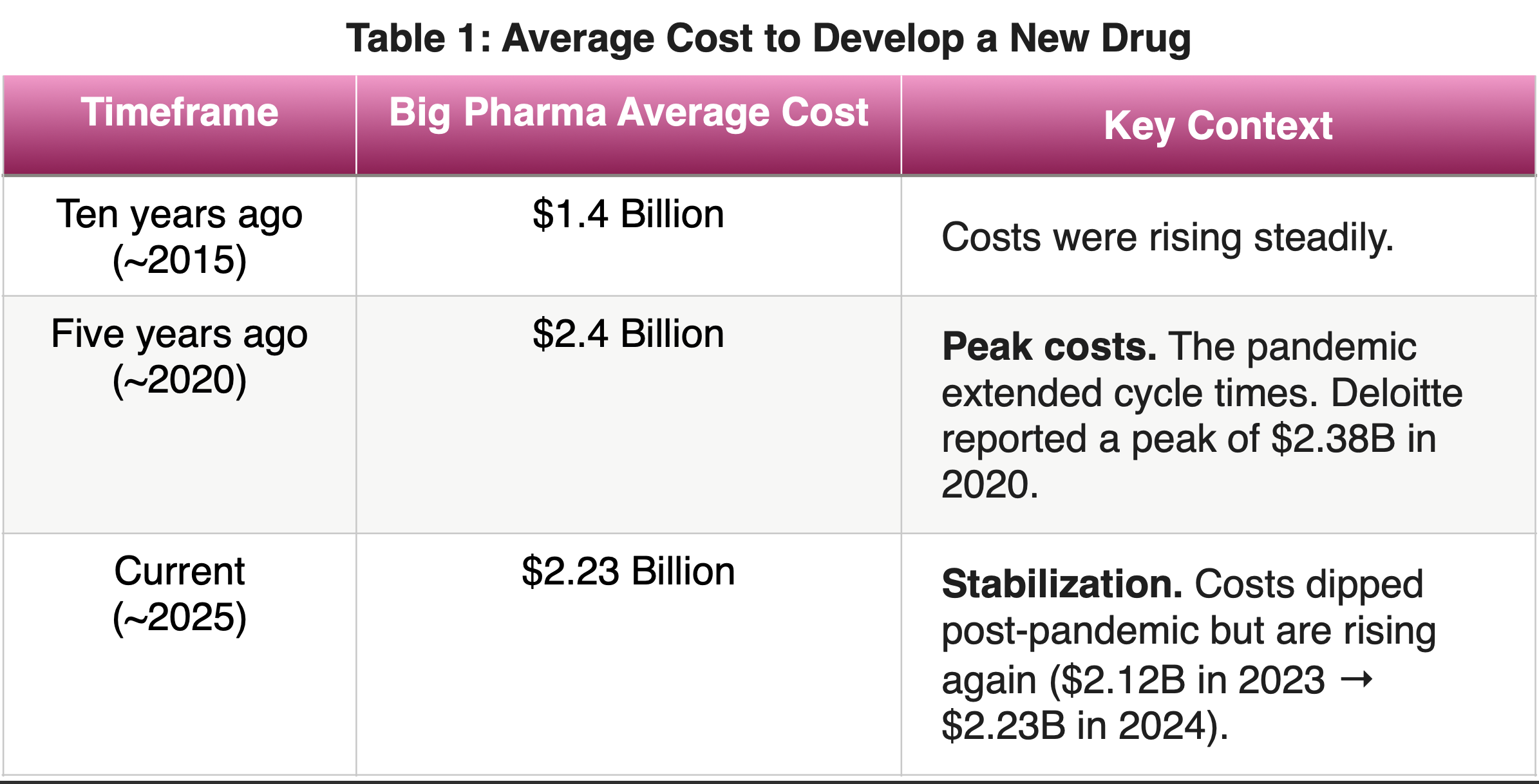
Why isn’t the price tag dropping?
Between 2015 and 2020, costs skyrocketed. We saw a “race for complex biologics” and immense competition for clinical trial sites. Even though AI is speeding up the discovery of molecules, the “Cycle Time”—the total time from lab to patient—has barely budged despite the “AI frenzy”. According to reports from McKinsey and IQVIA, the “Physical Bottleneck” is the culprit. We are finding molecules in weeks using AI, but we are still stuck in a decade-long clinical trial system that relies on human data and physical trial sites (Figure 1).
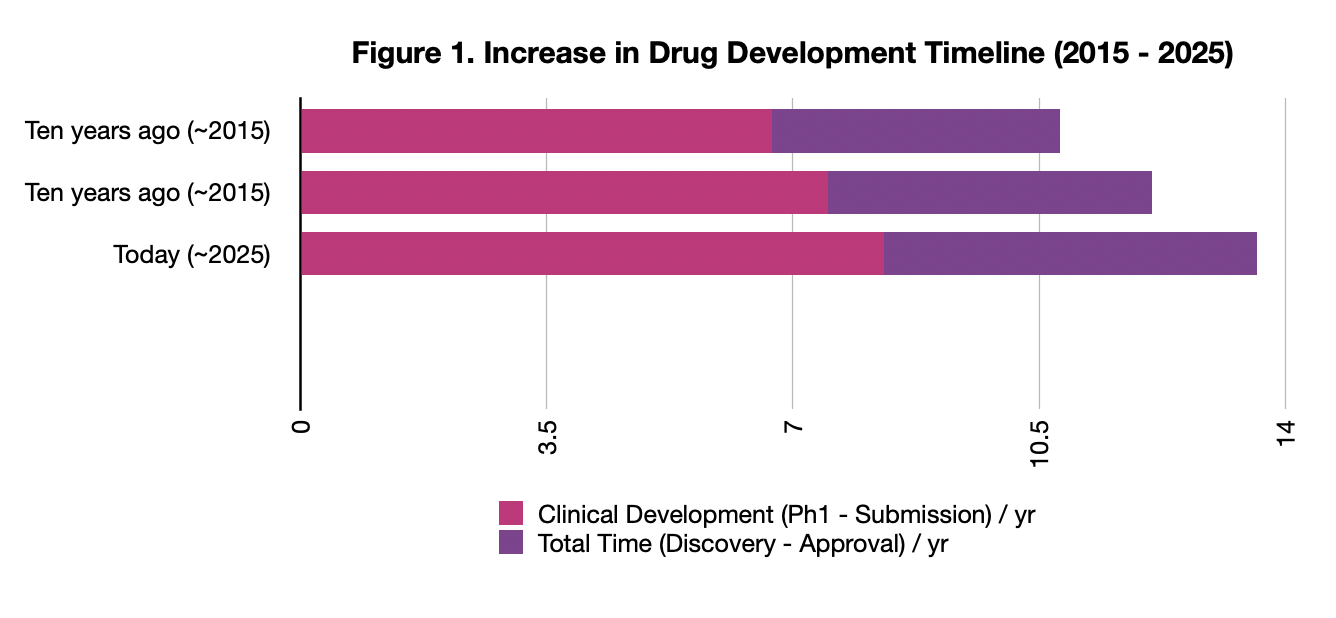
The AI “Inflection Point”
According to 2025 reports from the World Economic Forum and McKinsey, the industry has shifted from asking “Does AI work?” to “Where is AI best used?” Nevertheless, the consensus is that:
AI is a speed and safety tool, but human clinical trials remain the ultimate, un-skippable test for whether a drug truly heals a patient.
Right now, most companies are using AI as a “Convenient tool” or for “human augmentation”. We are accelerating the start of the race while the finish line remains 10 miles away, guarded by prolonged clinical development. Many companies are settling for being ‘AI savvy’ rather than ‘AI transformative’. But for visionary leaders, the goal isn’t just to have an AI department—it’s to solve the ROI Paradox.
In our next episode
We’re going to discuss the “Survivorship Bias.” We’ll compare the top 5 AI-designed drugs of 2025 and see who is actually winning—and who just has a great PR team.
Disclaimer:
The information provided in this article is for general informational and educational purposes only. It is not intended as, and should not be construed as, professional business, legal, investment, or medical advice. Before making any strategic decisions, you should consult with a qualified professional. Calxera makes no representation or warranty of any kind, express or implied, regarding the accuracy, adequacy, validity, reliability, availability, or completeness of any information on this site.