MiRNA: Nobel Glory, Clinical Failure—Unpacking the Crisis of the 'Next Generation' RNA Drug
November 17, 2025

Last year, the Nobel Prize in Physiology or Medicine was awarded to Victor Ambros and Gary Ruvkun for their foundational discovery of how microRNAs (miRNAs) influence gene expression. Decades of effort have since been dedicated to elucidating how genetic pathways are regulated by miRNAs and how abnormal miRNA expression leads to disease. However, developing miRNA-based therapies to reverse disease symptoms has proven to be an uphill battle. Unlike other nucleic acid therapies, such as small interfering RNA (siRNA) and antisense oligonucleotides (ASOs), there is currently no miRNA-based drug approved by the FDA. Furthermore, the number of unique miRNA therapeutic candidates that have entered clinical trials (Phase I, II, or III) remains relatively small compared to these other established modalities.
What is Slowing Down miRNA Therapies?
The development of miRNA therapies faces several key hurdles:
-
Delivery and Specificity Challenges MiRNA therapies suffer from the major challenges common to any RNA-based treatment: delivery problems and toxicity leading to adverse immune responses. A primary concern is that miRNAs inherently possess lower specificity compared to an siRNA, which is typically designed to target a single sequence. This broader targeting profile makes functional validation time-consuming and expensive. Note: It is crucial to distinguish between miRNA mimics (designed to restore function) and miRNA inhibitors (designed to block function), as their molecular design strategies are fundamentally different.
-
Complexity of Mechanism of Action (MOA) Beyond the initial challenges in design and screening, demonstrating a clear and reproducible mechanism of action (MOA) for a particular miRNA pathway is often a complicated task. Elucidating precisely how phenotypic restoration is achieved subsequently impacts critical development steps, including pharmacokinetics measurement, toxicity de-risking, and the design of effective translational biomarker strategies. Moreover, finding an efficient delivery method remains a lingering question, even as recent progress in delivery engineering begins to show promise in targeting certain tissues.
Is miRNA a Hidden Goldmine?
We believe the answer is a resounding YES. A growing number of companies are now boldly embarking on their miRNA therapeutic journey. We reason that this resurgence is largely driven by the ever-growing demand for novel therapeutic targets. Looking at investment and business development (BD) activity, more biopharma companies are taking significant steps into miRNA therapeutic discovery and development.
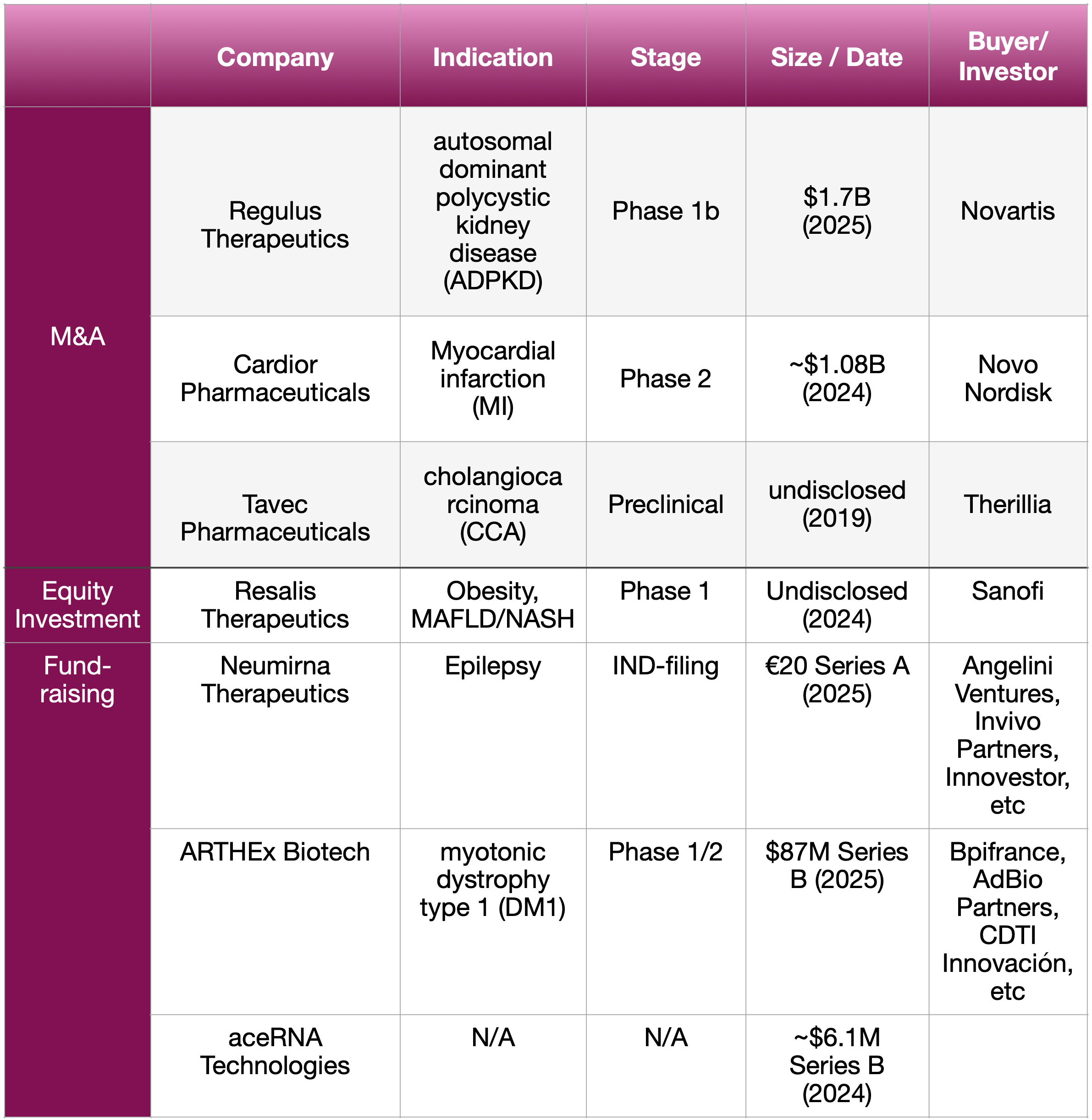
As our knowledge of RNA therapeutics continues to grow and mature, it’s perhaps time to abandon the old stereotypes about the difficulties associated with miRNAs. We predict that more significant business deals and technology breakthroughs concerning miRNAs will emerge in the near future —— Will you join the miRNA adventure?
Reference: What will it take to get miRNA therapies to market?. Nat Biotechnol 42, 1623–1624 (2024). https://doi.org/10.1038/s41587-024-02480-0
Disclaimer:
The information provided in this article is for general informational and educational purposes only. It is not intended as, and should not be construed as, professional business, legal, investment, or medical advice. Before making any strategic decisions, you should consult with a qualified professional. Calxera makes no representation or warranty of any kind, express or implied, regarding the accuracy, adequacy, validity, reliability, availability, or completeness of any information on this site.